BBB4B897-AE6F-41B2-9016-8E5B34C2F9FF.jpeg - 1 gigatonne Gt =1 000 000 000 000 000 g 1 megatonne Mt =1 000 000 000 000 g 1 tonne t =1 000 000 g 1 | Course Hero

Josh Rosen on Twitter: ". @Google Just FYI, Google search boldly states that 1 liter of water has a mass of 1,000 kg because the featured snippet later mentions 1,000 liters is

End-use emissions of biogas in different scenarios (g per 1 tonne of... | Download Scientific Diagram
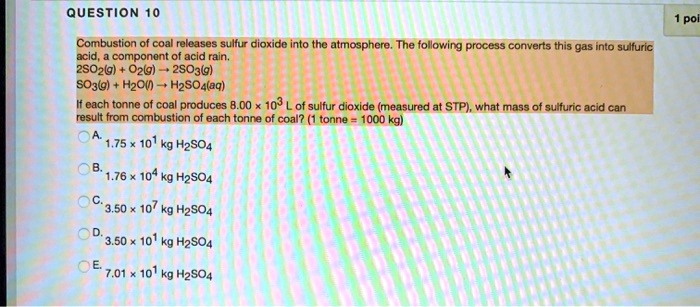
SOLVED: QUESTiOn 10 Combustion coal releases sulfur dioxide into thc atmosphere The following process converts this gas into sulfuric ocid componont of acid rain: 2SO2/g) Oz/g) 2SO3lg) SO3/g) Hzou H2SO4laq) each tonne

What is the ∆H value for this thermo chemical equation? Full question in the description box below. | Socratic
![SOLVED: (ii) Calculate the maximum volume, in dm^3, of chlorine gas at rtp that can be obtained from 23.4 tonnes of molten sodium chloride. [1. tonne .=10^6 g] [Mr. of .NaCl=58.5] [molar SOLVED: (ii) Calculate the maximum volume, in dm^3, of chlorine gas at rtp that can be obtained from 23.4 tonnes of molten sodium chloride. [1. tonne .=10^6 g] [Mr. of .NaCl=58.5] [molar](https://cdn.numerade.com/ask_images/3d073add-42ef-4e80-a854-08dbf5f54014.jpg)


![Land-Rover 101 FC -1-tonne- ambulance [Luxembourg Army] | Flickr Land-Rover 101 FC -1-tonne- ambulance [Luxembourg Army] | Flickr](https://live.staticflickr.com/7323/9968337105_ee2435df66_b.jpg)