
The Glaser–Hay Reaction: Optimization and Scope Based on 13C NMR Kinetics Experiments - Vilhelmsen - 2013 - European Journal of Organic Chemistry - Wiley Online Library
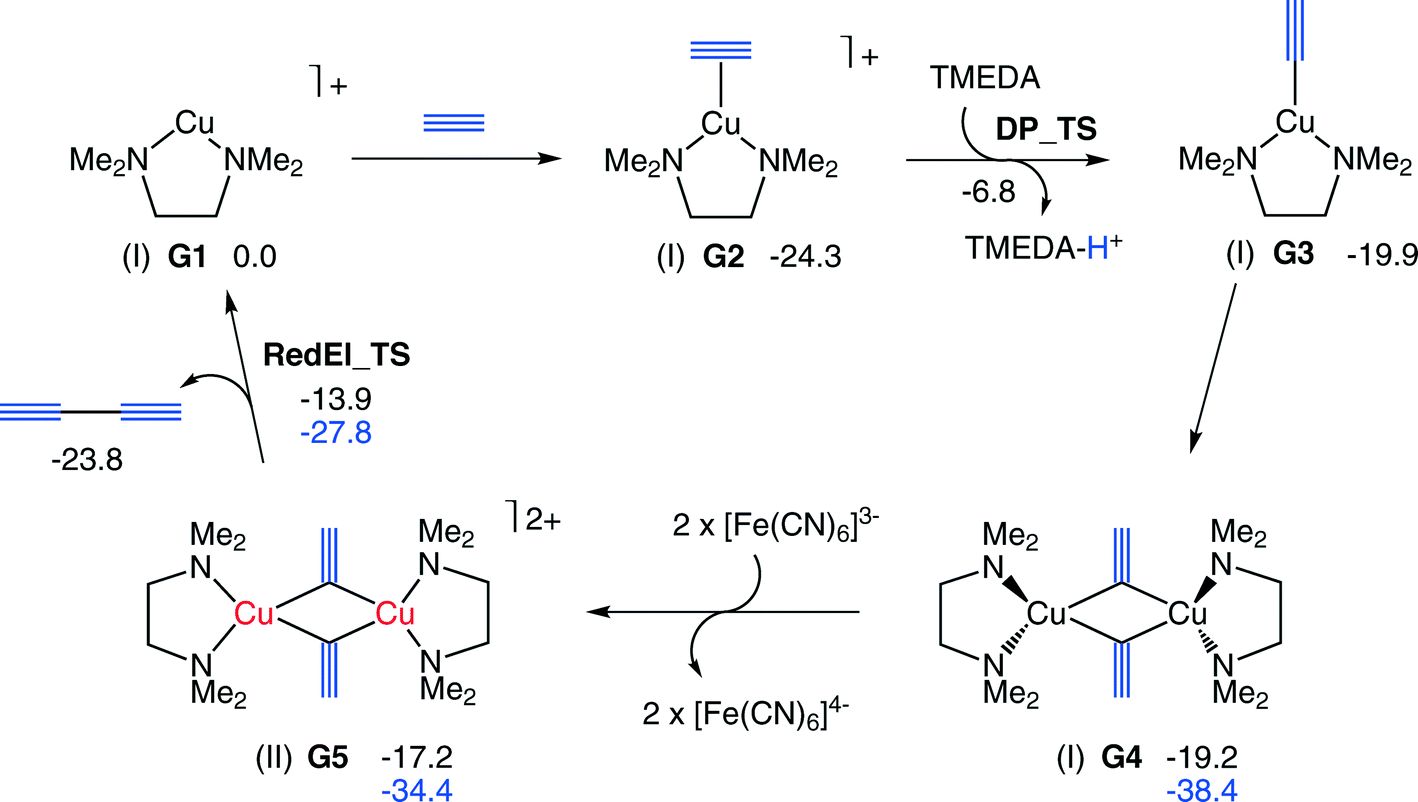
Toward a mechanistic understanding of oxidative homocoupling: the Glaser–Hay reaction - Catalysis Science & Technology (RSC Publishing) DOI:10.1039/C4CY00322E

The Glaser–Hay Reaction: Optimization and Scope Based on 13C NMR Kinetics Experiments - Vilhelmsen - 2013 - European Journal of Organic Chemistry - Wiley Online Library
![Cu(I)-azidopyrrolo[3,2-d]pyrimidine Catalyzed Glaser–Hay Reaction under Mild Conditions | ACS Organic & Inorganic Au Cu(I)-azidopyrrolo[3,2-d]pyrimidine Catalyzed Glaser–Hay Reaction under Mild Conditions | ACS Organic & Inorganic Au](https://pubs.acs.org/cms/10.1021/acsorginorgau.1c00015/asset/images/large/gg1c00015_0001.jpeg)
Cu(I)-azidopyrrolo[3,2-d]pyrimidine Catalyzed Glaser–Hay Reaction under Mild Conditions | ACS Organic & Inorganic Au

The Glaser–Hay Reaction: Optimization and Scope Based on 13C NMR Kinetics Experiments - Vilhelmsen - 2013 - European Journal of Organic Chemistry - Wiley Online Library
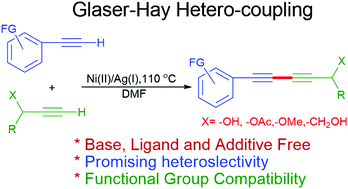
Glaser–Hay hetero-coupling in a bimetallic regime: a Ni(ii)/Ag(i) assisted base, ligand and additive free route to selective unsymmetrical 1,3-diynes - Chemical Communications (RSC Publishing)












