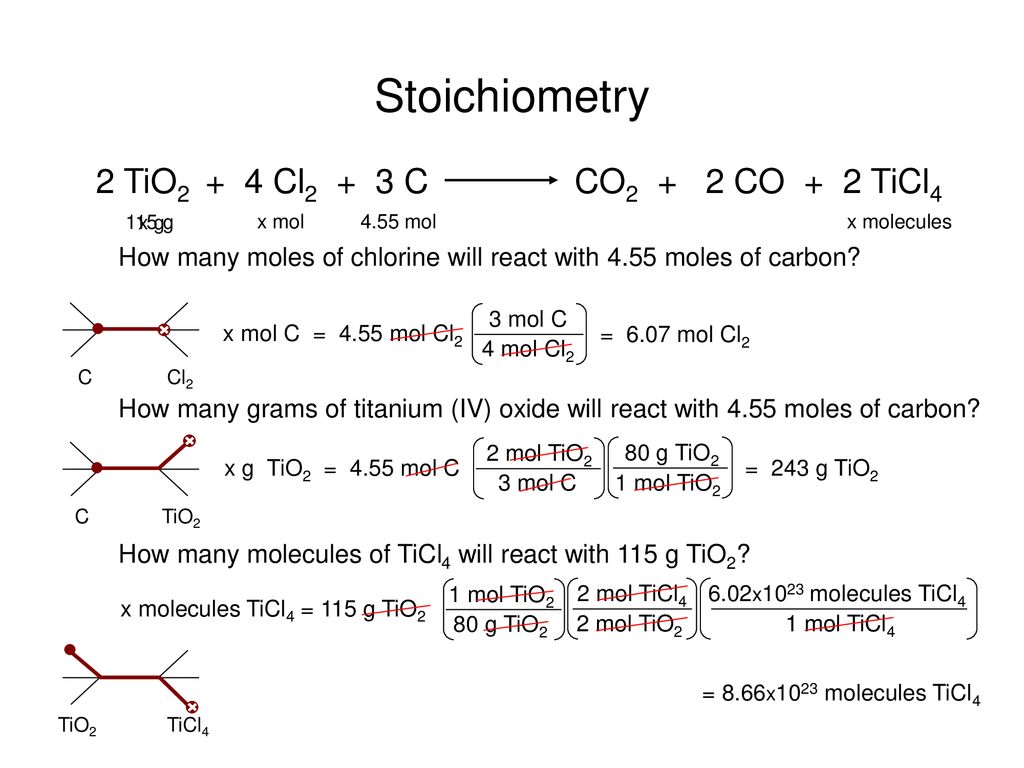
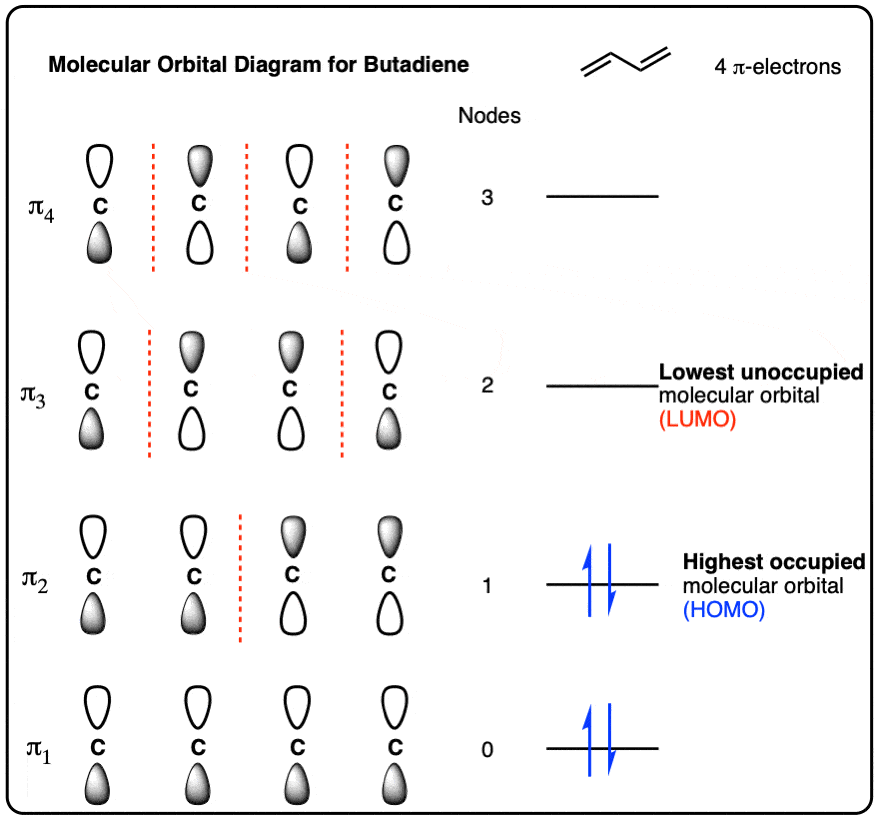
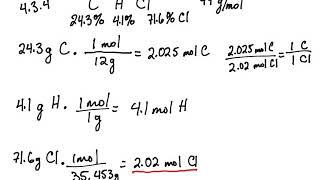Draw the complete molecular orbital diagram for C^+_2 (form the molecular orbital diagram from the combination of a neutral C and a cationic C^+). | Homework.Study.com

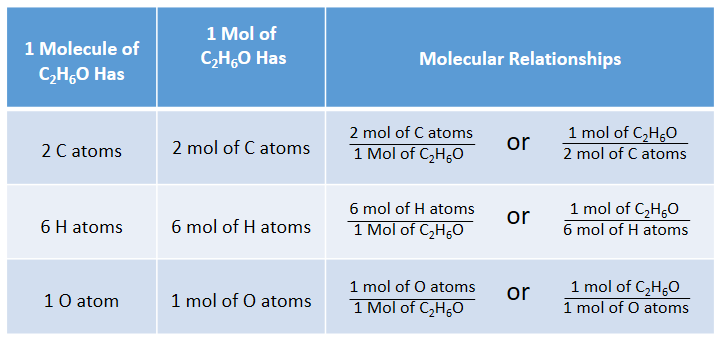
Empirical and molecular formulas for compounds that contain only carbon and hydrogen (C a H b ) or carbon, hydrogen, and oxygen (C a H b O c ) can be determined with a process called combustion analysis. The steps for this procedure are

Empirical and molecular formulas for compounds that contain only carbon and hydrogen (C a H b ) or carbon, hydrogen, and oxygen (C a H b O c ) can be determined with a process called combustion analysis. The steps for this procedure are
42. 1 mole of 'A', 1.5 mole of 'B' and 2 mole of 'C' are taken in a vessel of volume one litre . At equilibrium concentration of C is 0.5 mole/L.




:max_bytes(150000):strip_icc()/vitamin-c-molecular-model-483948223-582c8a523df78c6f6a473f1c.jpg)