
Закончите уравнение и определите тип химической реакции •Na2O + H2O •Na2SiO3 + H2SO4 •Al + - Школьные Знания.com

Stability diagrams for some minerals in the systems Na2–Al2O3–SiO2–H2O... | Download Scientific Diagram

Chemical Reactions Balancing Chemical Reactions. Chemical Reactions Objectives List three observations that suggest that a chemical reaction has taken. - ppt download

EDTA Disodium salt solution for 1000 ml volumetric solution c(Na2-EDTA X 2 H2O) 0.1 mol/l (0.2N) - Th. Geyer

Complete and balance the following equations :(a) Na + O2 → (b) Na2O + H2O → (c) Fe(s) + H2O(g) red heat (d) Cu(NO3)2 (aq) + Zn(s) →
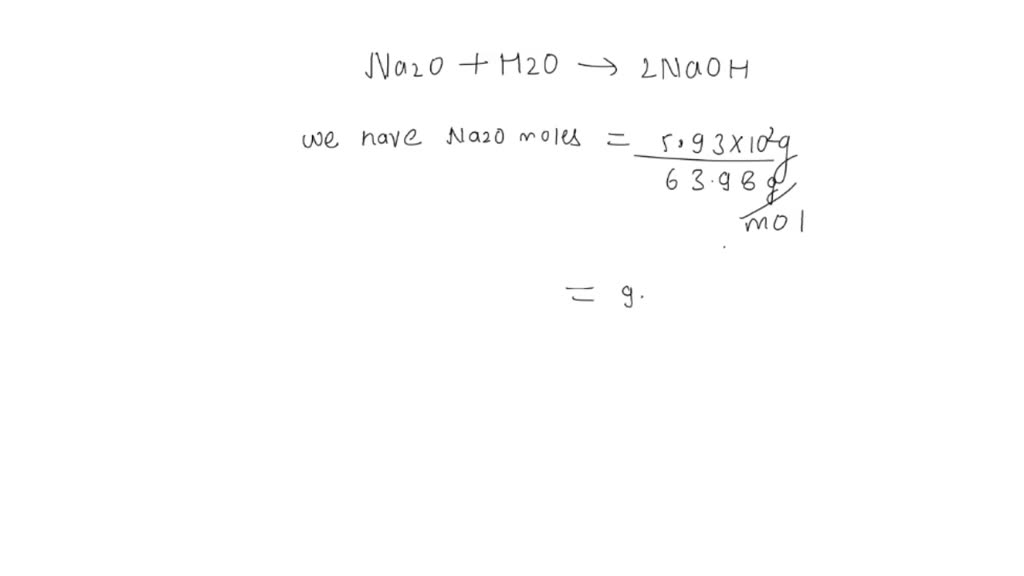
SOLVED: 3. Given the following unbalanced reaction: Na2O + H2O —> NaOH How many grams of NaOH is produced from 5.93 x 102 grams of Na2O?
![Effect of Oxygen on Ammonothermal Synthesis: Example of Na2[Zn(NH2)4] ⋅ (NH3)x and Na2[Zn(NH2)4] ⋅ (H2O)x - Kunkel - 2021 - European Journal of Inorganic Chemistry - Wiley Online Library Effect of Oxygen on Ammonothermal Synthesis: Example of Na2[Zn(NH2)4] ⋅ (NH3)x and Na2[Zn(NH2)4] ⋅ (H2O)x - Kunkel - 2021 - European Journal of Inorganic Chemistry - Wiley Online Library](https://chemistry-europe.onlinelibrary.wiley.com/cms/asset/6d0a2979-5094-445c-a1c5-5dbc03a1765e/ejic202100721-toc-0001-m.jpg)


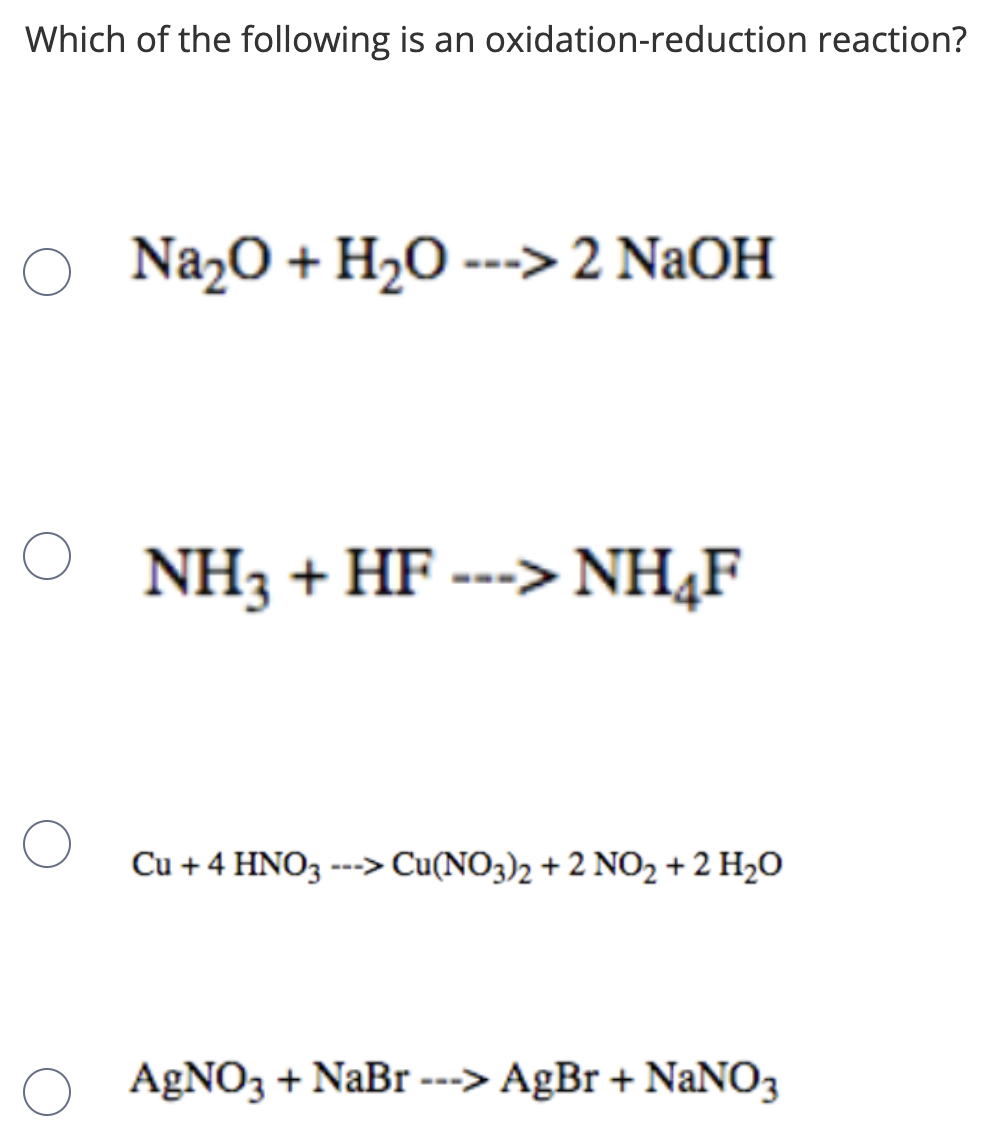




![a) Exemplary structure of the complex Na2[Hf2(dpta)2] 7.5H2O .... | Download Scientific Diagram a) Exemplary structure of the complex Na2[Hf2(dpta)2] 7.5H2O .... | Download Scientific Diagram](https://www.researchgate.net/publication/352533926/figure/fig2/AS:1036268796379136@1624077142150/a-Exemplary-structure-of-the-complex-Na2Hf2dpta2-75H2O-05EtOH-b-2D-1-H-13-C.png)